Infectious Diseases
The fight against infectious diseases has been one of Meiji's major social contributions, from our early manufacture of penicillin drugs beginning in 1946 to the development of the world's first and only oral carbapenem antibiotic. We also produce generic antibacterial agents for systemic use, while our vaccine products allow us to prevent infectious diseases, not only treat them.
Antibiotics
The development of antibacterial drugs has been a core strength of Meiji for more than 70 years. We manufacture a range of antimicrobial agents for respiratory and other infections.
Vaccines
Meiji produces vaccines for influenza, hepatitis, Japanese encephalitis, rabies, and a quadruple vaccine containing antigens of Bordetella pertussis and others. We are also developing vaccines for new coronaviruses. See vaccines for details.
Detection Kits
Rapid diagnosis enables better use of microbial drugs and allows us to predict and curb the spread of disease. Meiji sells rapid diagnosis kits for Influenza and COVID-19.
Antiseptics
The best way to stop infections is to prevent their spread. Our gargles and hand sanitizers contain povidone-iodine, an antiseptic ingredient that uses the oxidizing action of iodine.


-
Infectious Disease Control
The stable supply of antimicrobial agents is a national security issue, as manufacturing delays overseas can quickly impact on domestic medical care. Ten antibacterial drugs have been specified by academic societies as clinically important "Key Drugs" in Japan. Meiji has a large market share for several of these key drugs and is looking at ways to strengthen its supply chains to reduce any risks to the general population.
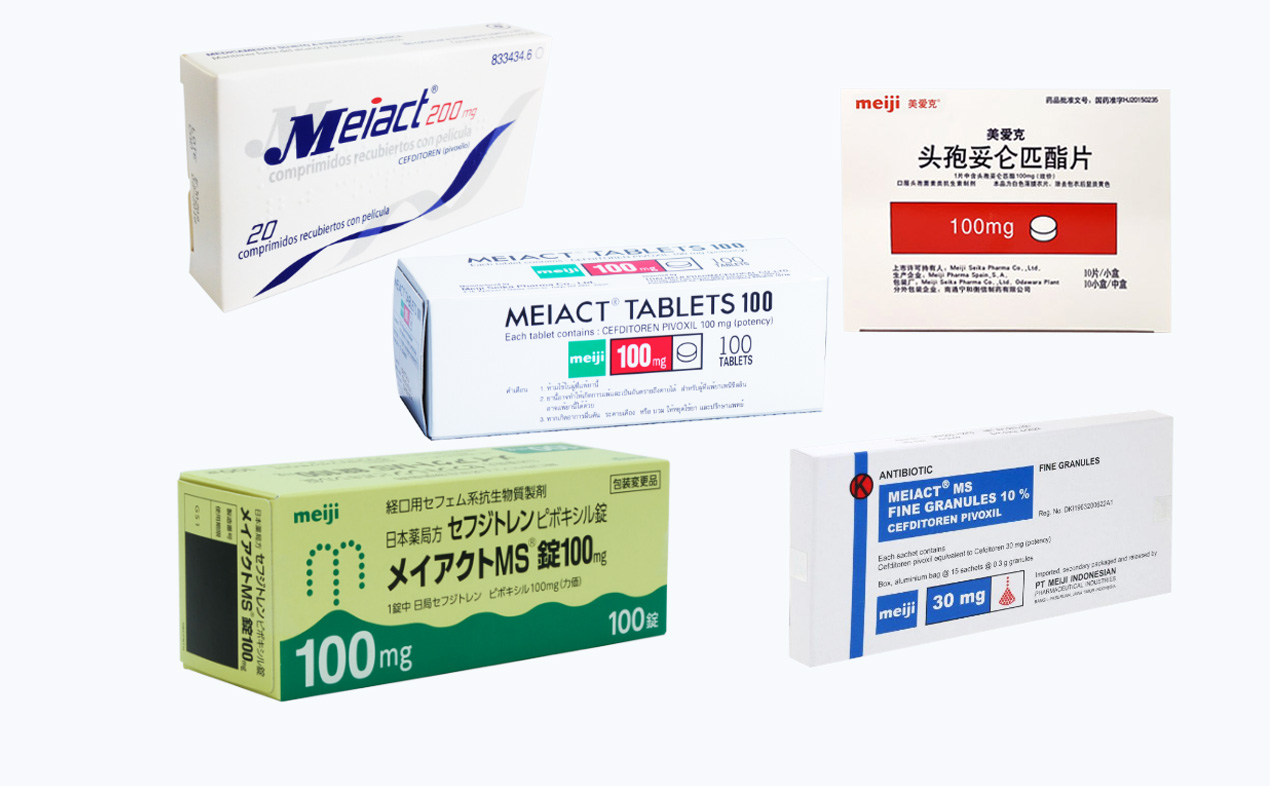
Meiact: A Leading Global Antibiotic
Meiact, an antibiotic drug first approved in Japan in 1994, has become widely prescribed globally. Later clinical trial data compiled by Meiji Seika Pharma found Meiact to be effective against newly resistant bacteria, which attracted a great deal of attention at the time. Meiact has a reputation for safety, which has made it a popular prescription for children. It is now approved for use in many parts of the world including Europe and Asia.
Find out more from our wellness stories
The Hidden Health Crisis of AMR
Meiji is tackling a health crisis the pharmaceutical industry has largely ignored: the spread of antibiotic-resistant bacteria.
Areas of Focus
Have a question?
Contact us here.
CONTACT US
Meiji Seika Pharma Co., Ltd.
Based in Japan, we are a leading developer and manufacturer of antibacterial drugs, vaccines and biotech solutions, with a history of over 75 years.
Location
- Headquarters
2-4-16 Kyobashi, Chuo-ku, Tokyo104-8002 Japan